Medical Devices
Medtronic’s OmniaSecure Defibrillation Lead Gains FDA Approval for Conduction System Pacing
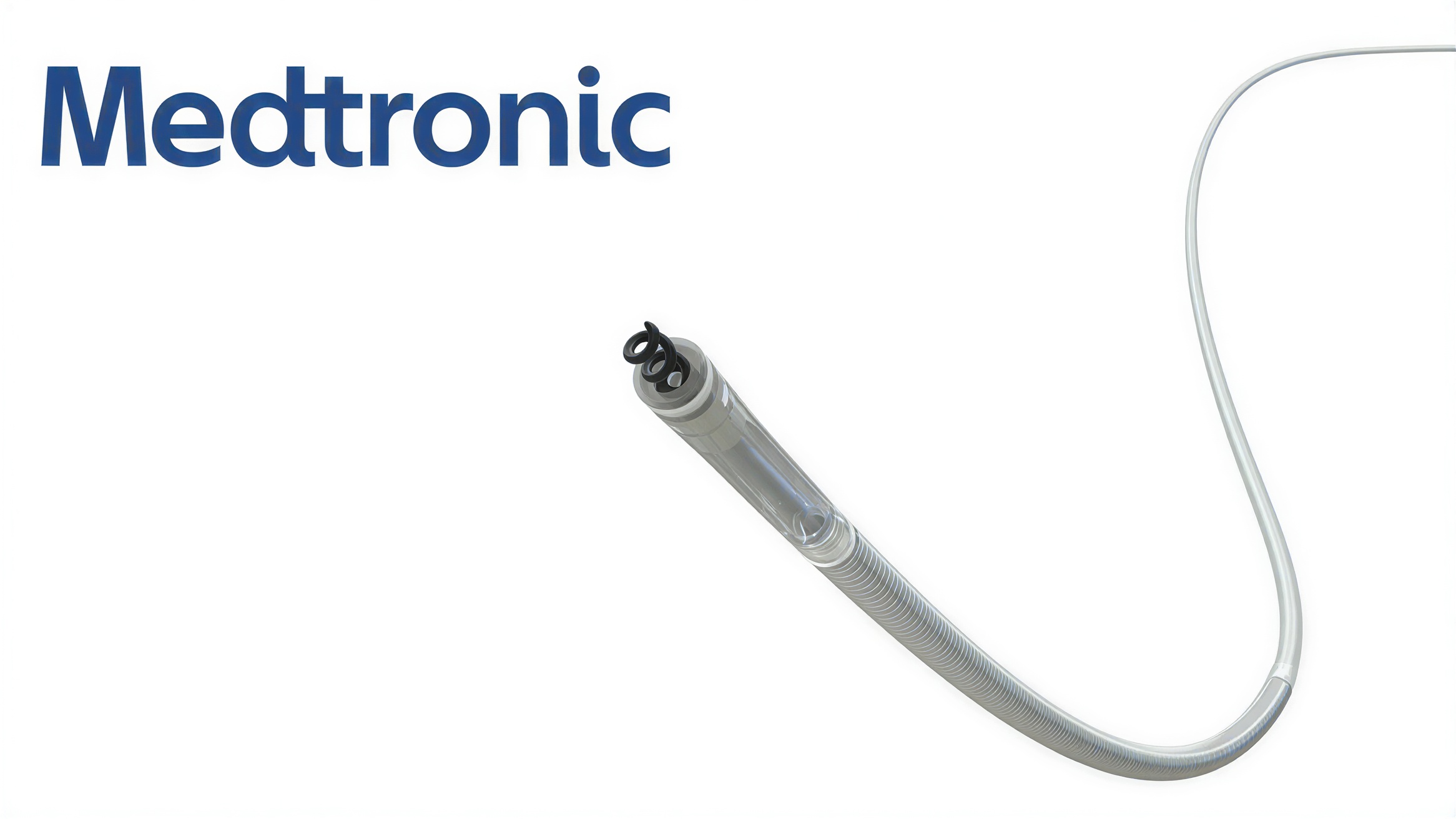
First defibrillation lead approved for left bundle branch pacing to support more physiologic cardiac therapy
Medtronic plc has received U.S. Food and Drug Administration (FDA) approval for an expanded indication of its OmniaSecure™ defibrillation lead, enabling its placement in the left bundle branch (LBB) area for conduction system pacing (CSP). This approach closely mimics the heart’s natural electrical activity, supporting more physiologic pacing.
The approval also allows use in left bundle branch optimized cardiac resynchronization therapy (LOT-CRT), a novel approach that combines CSP with left-ventricular pacing to improve patient outcomes.
The OmniaSecure defibrillation lead connects to implantable cardioverter-defibrillators (ICDs) or cardiac resynchronization therapy defibrillators (CRT-Ds) to treat life-threatening arrhythmias, including ventricular tachyarrhythmias, ventricular fibrillation, and bradyarrhythmias. It is the first defibrillation lead approved for placement in the LBB area and is currently the smallest diameter defibrillation lead available, measuring 4.7 French (1.66 mm). It is also approved for use in adults and adolescent patients aged 12 years and above.
Built on the foundation of the Medtronic SelectSecure™ Model 3830 pacing lead, the OmniaSecure lead features a lumenless design that enhances durability and allows catheter-based delivery for precise placement in both the right ventricle and the LBB area. By leveraging the heart’s natural conduction pathways, CSP helps deliver therapy while reducing the risk of complications associated with conventional pacing methods.
"Conduction system pacing is a rapidly growing therapy for patients who need a pacemaker. Now, patients who require a defibrillator and pacing have an option that can safely deliver life-saving defibrillation therapy and activate the heart's natural electrical system to enable a more synchronous, physiologic pattern," said Trevor Cook, vice president and general manager of the Defibrillation Solutions business, which is part of the Cardiac Rhythm Management operating unit at Medtronic. "This approval underscores the versatility of the OmniaSecure defibrillation lead and supports its use across a variety of implant approaches to best serve a broad range of patients."
ICDs and CRT-Ds remain the gold standard for preventing sudden cardiac arrest. For patients requiring both pacing and defibrillation therapy, the OmniaSecure lead offers a combined solution that supports physiologic pacing through the LBB area when used with compatible Medtronic devices such as the Cobalt™ and Crome™ systems. Patients indicated for CRT may also benefit when the lead is used in biventricular or LOT-CRT configurations.
FDA approval was supported by data from the global LEADR LBBAP trial, which demonstrated strong safety and effectiveness outcomes. Study results showed 100% defibrillation success at implant and a low major complication rate of 2.1% at three months when the lead was placed in the LBB area.
"The robust body of clinical evidence from the LEADR LBBAP study supports the OmniaSecure defibrillation lead as a safe and effective way to activate physiologic pacing through the left bundle branch area or in applications like LOT-CRT, while also demonstrating reliable defibrillation success," said Pugazhendhi Vijayaraman, M.D., cardiac electrophysiologist at Geisinger Wyoming Valley Medical Center and principal investigator of the LEADR LBBAP study.
The approval further expands Medtronic’s portfolio of conduction system pacing solutions, including the SelectSecure Model 3830 pacing lead, the C320LBB delivery catheter, and the 5944RL rotatable connector, all designed to enhance procedural precision and long-term performance.
The OmniaSecure defibrillation lead was commercially launched in the United States in January 2026 following earlier approval for placement in traditional right ventricular locations.
The approval also allows use in left bundle branch optimized cardiac resynchronization therapy (LOT-CRT), a novel approach that combines CSP with left-ventricular pacing to improve patient outcomes.
The OmniaSecure defibrillation lead connects to implantable cardioverter-defibrillators (ICDs) or cardiac resynchronization therapy defibrillators (CRT-Ds) to treat life-threatening arrhythmias, including ventricular tachyarrhythmias, ventricular fibrillation, and bradyarrhythmias. It is the first defibrillation lead approved for placement in the LBB area and is currently the smallest diameter defibrillation lead available, measuring 4.7 French (1.66 mm). It is also approved for use in adults and adolescent patients aged 12 years and above.
Built on the foundation of the Medtronic SelectSecure™ Model 3830 pacing lead, the OmniaSecure lead features a lumenless design that enhances durability and allows catheter-based delivery for precise placement in both the right ventricle and the LBB area. By leveraging the heart’s natural conduction pathways, CSP helps deliver therapy while reducing the risk of complications associated with conventional pacing methods.
"Conduction system pacing is a rapidly growing therapy for patients who need a pacemaker. Now, patients who require a defibrillator and pacing have an option that can safely deliver life-saving defibrillation therapy and activate the heart's natural electrical system to enable a more synchronous, physiologic pattern," said Trevor Cook, vice president and general manager of the Defibrillation Solutions business, which is part of the Cardiac Rhythm Management operating unit at Medtronic. "This approval underscores the versatility of the OmniaSecure defibrillation lead and supports its use across a variety of implant approaches to best serve a broad range of patients."
ICDs and CRT-Ds remain the gold standard for preventing sudden cardiac arrest. For patients requiring both pacing and defibrillation therapy, the OmniaSecure lead offers a combined solution that supports physiologic pacing through the LBB area when used with compatible Medtronic devices such as the Cobalt™ and Crome™ systems. Patients indicated for CRT may also benefit when the lead is used in biventricular or LOT-CRT configurations.
FDA approval was supported by data from the global LEADR LBBAP trial, which demonstrated strong safety and effectiveness outcomes. Study results showed 100% defibrillation success at implant and a low major complication rate of 2.1% at three months when the lead was placed in the LBB area.
"The robust body of clinical evidence from the LEADR LBBAP study supports the OmniaSecure defibrillation lead as a safe and effective way to activate physiologic pacing through the left bundle branch area or in applications like LOT-CRT, while also demonstrating reliable defibrillation success," said Pugazhendhi Vijayaraman, M.D., cardiac electrophysiologist at Geisinger Wyoming Valley Medical Center and principal investigator of the LEADR LBBAP study.
The approval further expands Medtronic’s portfolio of conduction system pacing solutions, including the SelectSecure Model 3830 pacing lead, the C320LBB delivery catheter, and the 5944RL rotatable connector, all designed to enhance procedural precision and long-term performance.
The OmniaSecure defibrillation lead was commercially launched in the United States in January 2026 following earlier approval for placement in traditional right ventricular locations.
Cardiac DevicesCardiology InnovationConduction System Pacing
Loading news...
PODCAST

Events & Summits
Host or sponsor events that place your brand at the forefront of healthcare impact.
Podcast Features
Engage a wider audience with thought leadership shared across VOH's podcast channels.
Brand Solutions
Take center stage at healthcare forums to spark conversations and share bold perspectives.
Events & Summits
Host or sponsor events that place your brand at the forefront of healthcare impact.
Podcast Features
Engage a wider audience with thought leadership shared across VOH's podcast channels.
Brand Solutions
Take center stage at healthcare forums to spark conversations and share bold perspectives.